Dortmund, 17th October 2022
Prof Dr Steven Verhelst, a project manager in the Proteomics research group, and Dr Daniel Krahn still have plenty of tricks in their bags. They want to combine knowledge from chemistry and biology at ISAS in order to combat cancer more efficiently and with fewer side effects than before. The focus of their work in the Proteomics research group is on active pharmaceutical substances whose targets they hope to adjust with new chemical tools for cancer therapy.
Cancer medicine has undergone tremendous development in the past decade, but some areas still require action or remain in the dark. For example, metastases are still the main cause of death in cancer patients. And while sufferers are fighting for their lives, other health complaints often come along, including side effects triggered by the drugs that are supposed to help. In some cases, they even hinder therapy or lead to concomitant diseases. The cardiovascular system suffers from the stress to which it is subjected in old age. In addition, cancer patients have a significantly elevated risk of stroke or heart failure, depending on the medication.
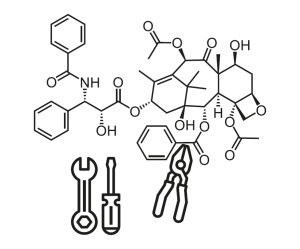
In the Proteomics research group, Prof Dr Steven Verhelst and Dr Daniel Krahn work on methods for optimising active substances (i.e. natural products such as Taxol) at the molecular level.
© ISAS
First hit the right target
The research project on late stage functionalisation (LSF) in chemical proteomics is concerned with so-called target effects in the body. Every active pharmaceutical substance has a target for which it has been developed. These targets can be structures such as enzymes, ion channels or receptor proteins that are involved in the development of a disease – in Verhelst and Krahn’s case, cancer cells – and should be destroyed. Although the molecules are very specific, there are structures (off-targets) to which they bind, even though they are not intended to do so at all. This off-target effect often manifests as an agonising side effect or concomitant disease for patients. “A better understanding of off-target effects could make cancer treatments safer and more tolerable than before. In this way, we could find targets for new drugs in future that stop or even prevent metastases,” Verhelst explains.

Prof Dr Steven Verhelst.
© ISAS
Better understanding of off-target effects could make cancer therapy safer & more tolerable.
A chemical toolbox
In 2021, scientists at ISAS began researching chemical tools that can be used to identify desirable and undesirable targets. They hope to use the knowledge gained from this development to improve drugs so that they bind only the right targets. In the long term, in addition to interdisciplinary collaboration with other teams, the two chemists will need a whole range of new chemical components (probes) with which to fill their toolboxes.
Twofold increased risk of cardiovascular disease in adults who survived cancer as children or adolescents compared with those without childhood or adolescent cancer.1
Like toolmakers, the researchers in the Proteomics research group use raw materials from which they aim to produce precise, customised tools. But these materials are active substances, consisting of tiny complex molecules, instead of large amounts of metal or plastic. These specific chemical probes could be used to further explore and improve existing cancer medications (such as those based on Taxol or other natural products).
Fight against »deadly daughters«
Although cancer therapies are very advanced today, patients still face one great danger – the spreading of tumours, i.e. cancer cells that break away from the tumour and migrate to other areas of the body where they form daughter tumours, referred to in medicine as metastases. Metastases are the cause of 90 per cent of cancer-related deaths here.
Verhelst and Krahn would like to use the chemical tools they have developed to further investigate the interaction between active substances and enzymes, for example. By doing so, they hope to uncover the hitherto little-studied networks within cancer cells. Communication in cancer cells plays a critical role in metastasis, and the two chemists suggest that metastases could be prevented once these networks are turned off or inhibited. If the Dortmund researchers, working with oncologists, succeed in better elucidating this interaction of tumour cells, it could result in a decisive advantage in the fight against cancer.
From old to new?
At this point, one might ask why this idea is needed. Why not design new drugs whose molecules bind the right targets and no off-targets? The answer is simple: On the one hand, according to Krahn, most of the off-targets are still unknown. And on the other hand, new molecules would again face unknown off-targets. Apart from that, the search for suitable active substances for new drugs takes several years. And the efforts do not promise success for a long time. “You may end up with dead ends by just excluding off-targets. And then, you might have a molecule that doesn’t dock to any off-targets but also doesn’t bind to the right targets,” Krahn says. The researcher calls such molecules »dead cows«. This comes from the saying, “You can’t milk a dead cow”.
The work on the chemical probes could help make the modification of an established active pharmaceutical substance easy, economical and, most importantly, quick. The chemical tools could be used to improve highly complex natural cancer therapy substances whose molecules currently bind unwanted off-targets or are unstable for the benefit of patients.

Dr Daniel Krahn.
© ISAS
1 Faber J et al., Burden of cardiovascular risk factors and cardiovascular disease in childhood cancer survivors: data from the German CVSS-study, European Heart Journal, Vol. 39, Nr. 17, 2018, Seiten 1555–1562, https://doi.org/10.1093/eurheartj/ehy026z.
(Cheyenne Peters / Sara Rebein)